Potassium Hydrogen Sulfate.
| Application Category | End Use. |
|---|---|
| Organic Reactions | Potassium Bisulfate is employed as a chemical catalyst and promoter in various organic reactions. It acts as an acid catalyst in the synthesis of esters and ethers. |
| Pharmaceuticals | Potassium Hydrogen Bisulfate acts as a chemical intermediate in the production of various pharmaceutical compounds. It is used as an excipient in pharmaceuticals. |
| Wine Making | Potassium HydrogenBisulfate is used as an additive in the preparation of potassium bitartrate (cream of tartar), which is used as a stabilizer in wine making and prevents the formation of tartrate crystals. |
| Flux Agent | It is used as a flux in the analysis of ores and siliceous compounds. |
| Analytical Chemistry | Potassium Hydrogen Bisulfate is used as a disintegrating agent (helps break down complex matrices) in sample preparation for various chemical analyses. |
| Food Additive | PotassiumBisulfate is used as a food preservative and acidity regulator in the food industry. It helps prevent the growth of bacteria, mold, and yeast, thus extending the shelf life of food products. ·It helps control the acidity of food, especially in beverages. INS Number: 515(II), E-Number: E515(II) |
| Metal Treatment | Potassium HydrogenBisulfate is used in the application of metal cleaning and pickling processes. |
| Dehydrating Agent | It is used as a dehydrating agent in the synthesis of certain compounds through various organic reactions. |
| Laboratory Reagent | Potassium Bisulfate is a common laboratory reagent used for various purposes, including pH adjustment and as a source of sulfate ions.
|
| Preparation of Other Chemicals | Potassium Hydrogen Bisulfate is used as a precursor in the production of potassium persulfate (a strong oxidizing agent in polymerization reactions and other industrial syntheses) through electrolysis. |
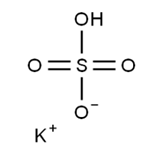
Potassium Hydrogen Sulfate Structure:
The chemical structure of potassium bisulfate consists of a potassium cation (K+) and a bisulfate anion (HSO4−). In the bisulfate ion, a central sulfur atom is bonded to four oxygen atoms. Two oxygen atoms are double-bonded to the sulfur atom, one negatively charged oxygen atom is single-bonded to the sulfur atom, and one oxygen atom, along with a hydrogen atom, is single-bonded to the sulfur atom.
Potassium Hydrogen Sulfate Production Process:
Industrially, the reaction of Potassium Chloride and Sulfuric Acid produces Potassium Bisulfate along with hydrogen chloride.
KCl + H2SO4 → KHSO4 + HCl
